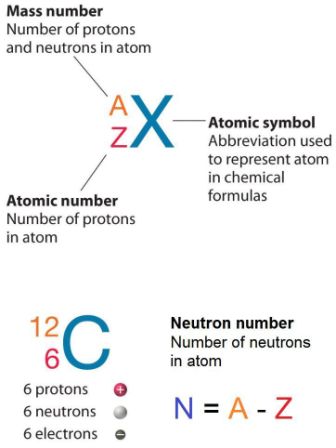
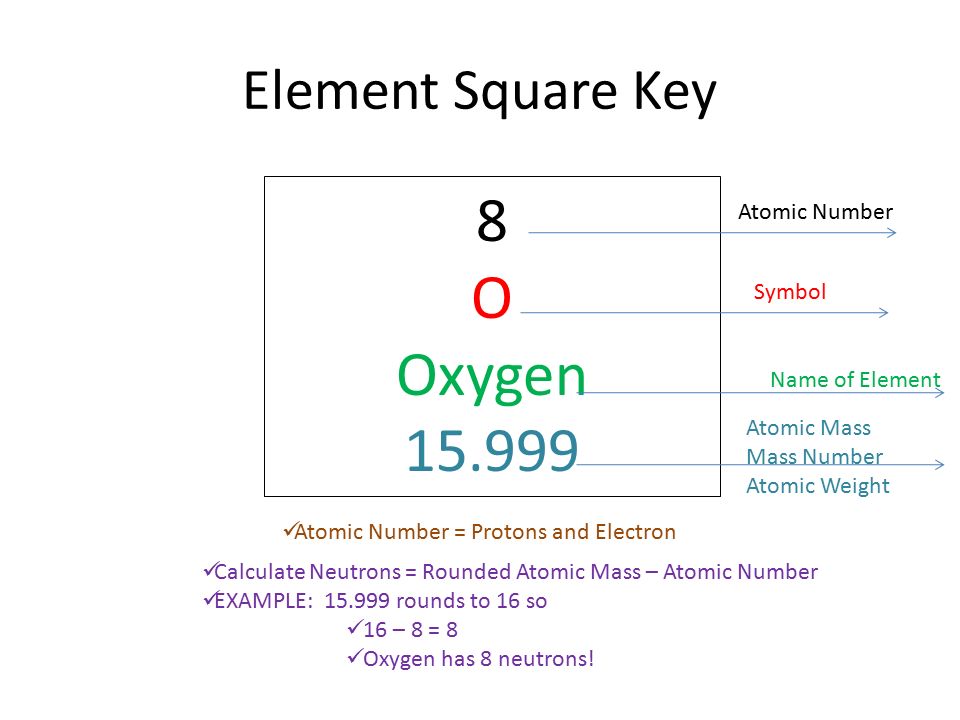
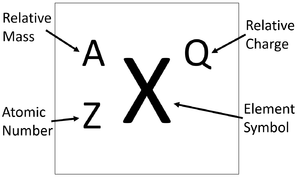
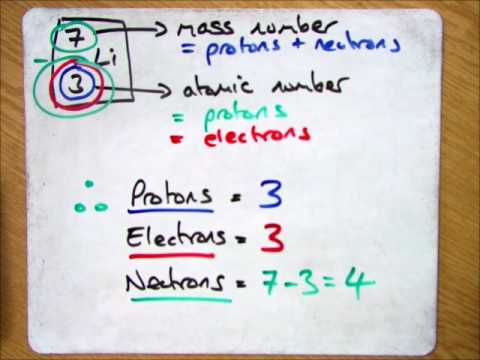
Atomic Number & Mass Number | How to Find the Atomic Mass Number - Video & Lesson Transcript | Study.com
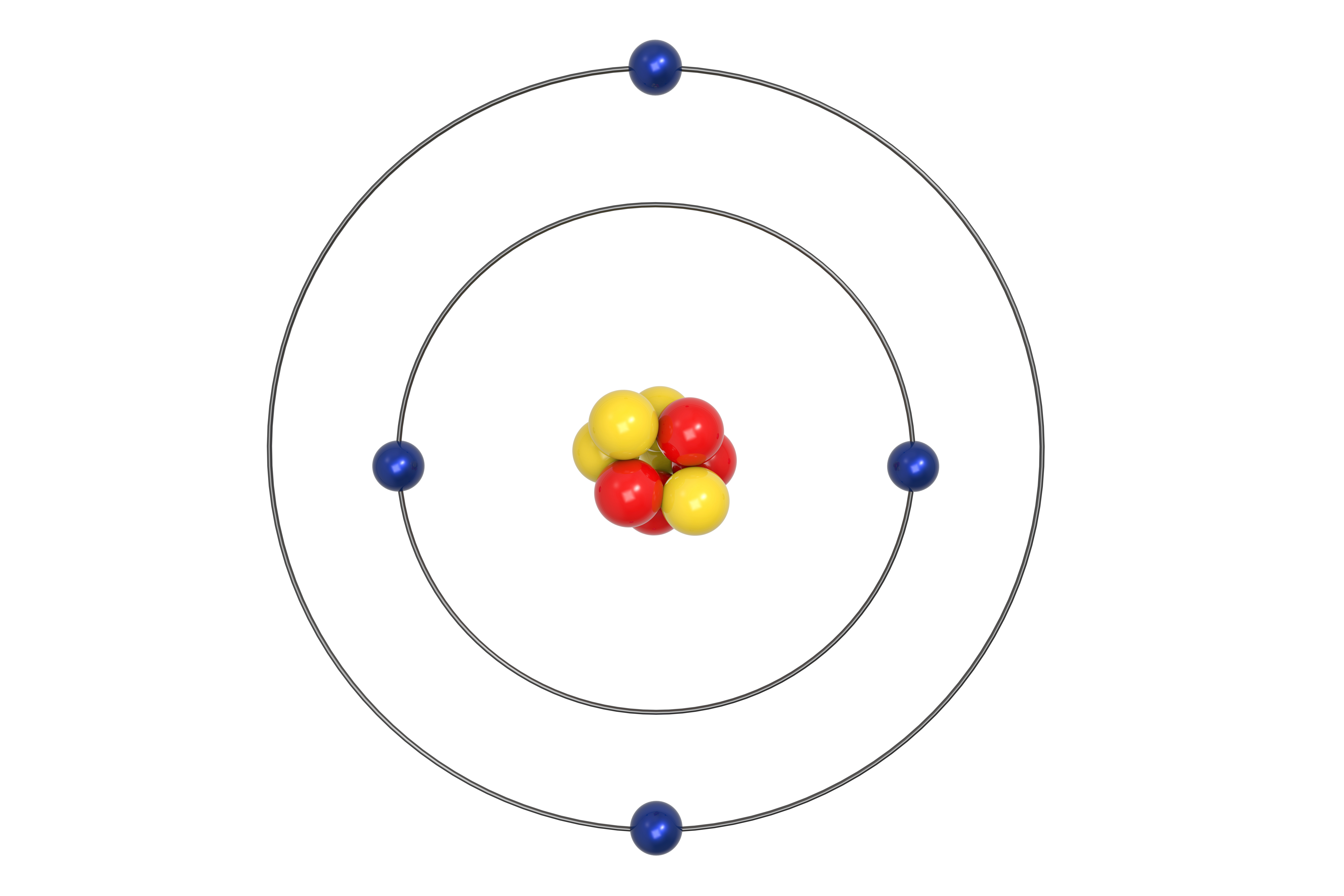
How can you determine the number of neutrons in an atom? - High School/Honors/AP® Chemistry Resources | Viziscience
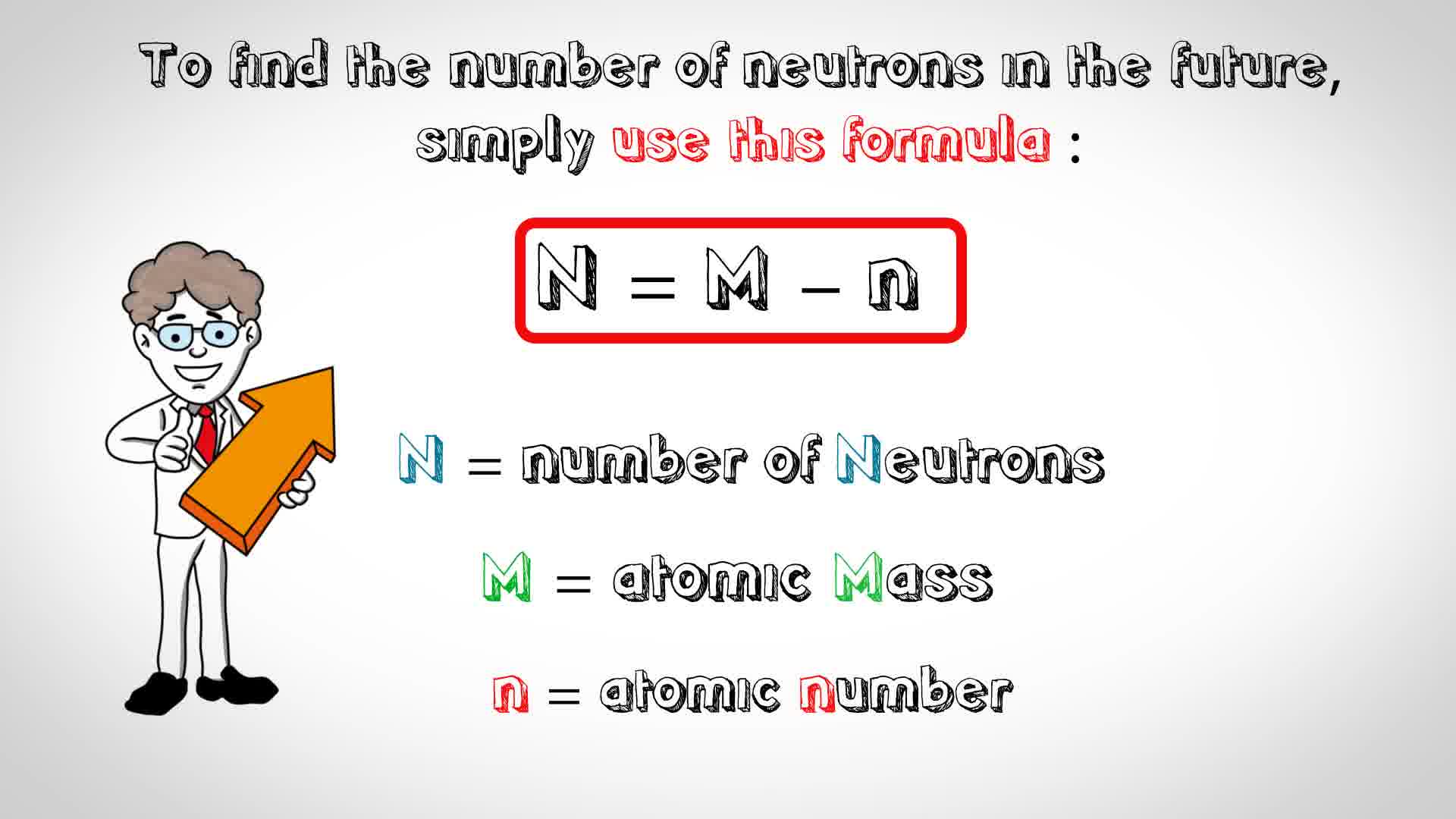
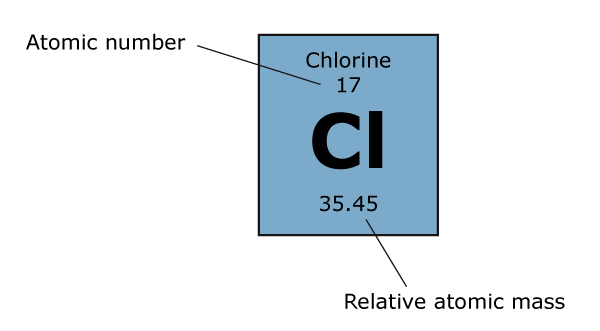
Zirconium has an atomic number of 40. How many neutrons are there in an isotope of zirconium-92? | Socratic
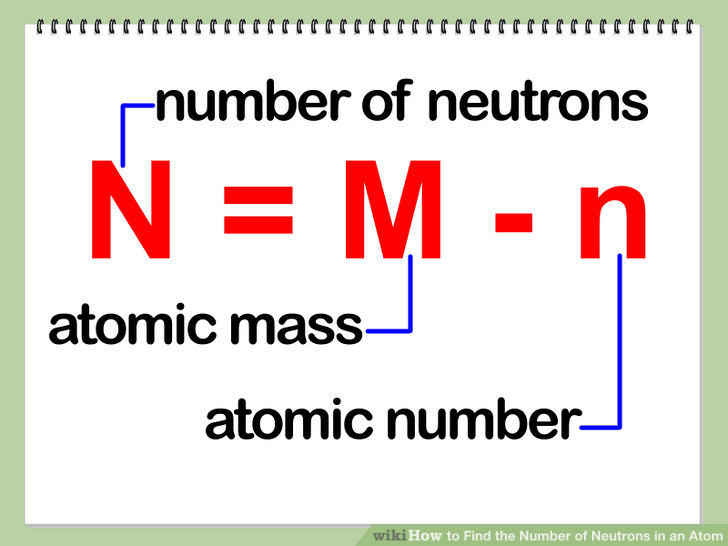
The mass number of a chromium atom is 52 and it has 24 protons. How many neutrons does this atom have? | Socratic
Calculate the number of electrons,protons,neutrons in 1.phosphorus atom. 2. Phosphate ion. MASS NO.=P=31, O=16 ATOMIC NO.P=15,O=8 along with explanation.